What is the internal resistance of a lithium battery?
When current flows through the inside of a battery, it encounters resistance, causing a decrease in the battery's operating voltage. This resistance is called the battery's internal resistance. Due to the battery's internal resistance, the terminal voltage is lower than the electromotive force and open-circuit voltage during discharge. During charging, the terminal voltage is higher than the electromotive force and open-circuit voltage. Battery internal resistance is an extremely important parameter of a chemical power source. It directly affects the battery's operating voltage, operating current, output energy, and power. For a practical chemical power source, the lower the internal resistance, the better.
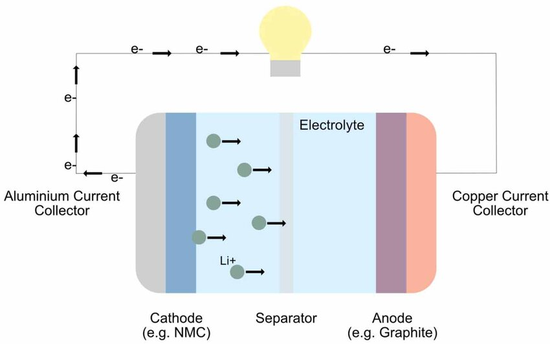
Battery internal resistance is not constant; it varies during discharge depending on the composition of the active material, the electrolyte concentration, the battery temperature, and the discharge time. Battery internal resistance includes the ohmic internal resistance (R₀) and the polarization internal resistance (Rf) exhibited by the electrodes during electrochemical reactions. The sum of these two is called the battery's total internal resistance (Rw).

Ohmic internal resistance is mainly composed of the internal resistance of electrode materials, electrolyte, separator, and the contact resistance of various components. It is related to the battery's size, structure, electrode forming method (e.g., paste-type and tubular electrodes in lead-acid batteries, and box-type and sintered electrodes in alkaline batteries), and assembly tightness. Ohmic internal resistance obeys Ohm's law.
Polarization internal resistance refers to the internal resistance caused by polarization between the positive and negative electrodes of a chemical power source during the electrochemical reaction. It is the sum of the resistances caused by electrochemical polarization and concentration
polarization.
Polarization internal resistance is related to the nature of the active material, electrode structure, and battery manufacturing process, especially closely related to the battery's operating conditions; discharge current and temperature have a significant impact. During high current density discharge, both electrochemical polarization and concentration polarization increase, potentially even causing passivation of the negative electrode, thus increasing polarization internal resistance. Low temperatures adversely affect electrochemical polarization and ion diffusion; therefore, the battery's polarization internal resistance also increases under low-temperature conditions. Therefore, the polarization resistance is not a constant, but changes with the discharge rate, temperature and other conditions.
The analytical expression for the internal resistance of a battery is as follows:

In the formula, bE (iₐ,τ,C) Iₐ⁻¹-Battery polarization internal resistance;
b-The coefficient of variation of the battery terminal voltage relative to the battery terminal voltage E under rated capacity conditions when the battery is charged and discharged with current Iₐ;
Rₑₗ(τ,C)-Resistance of the electrolyte;
Rₑ(C)-Electrode resistance. The electrolyte resistance Rₑₗ and electrode resistance Rₑ are inversely proportional to the instantaneous capacity of the battery;
iₐ, τ, C-Battery charging and discharging current, temperature, and capacity state at that time.
Battery internal resistance is relatively small and often negligible under many operating conditions. However, power batteries for electric vehicles often operate under high current and deep discharge conditions, resulting in a significant voltage drop caused by internal resistance. In this case, the impact of internal resistance on the entire circuit cannot be ignored.








